|
|
|

|
Suppliers for
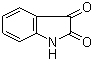
Isatin
|
Properties | | CAS |
91-56-5 | | Formula |
C8H5NO2 | | EINECS |
202-077-8 |
|
|
26 Registered suppliers
Molecular Formula: C8H5NO2
Molecular Weight: 147.13
Hazard Symbols: Xi,Xn
UN Number: 2811
WGKGermany: 1
PackingGroup: III
HS Code: 29337900
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
Molecular Formula: C8H5NO2
Molecular Weight: 147.1308
| Appearance | Orange-red Crystal
| | Assay | 95.0%min(first) 98.0%min(medicine)
| | Moisture | 1.0%max
| | M.P. | 198-204°C |
More details are to be found here
More details are to be found here
More details are to be found here
Purity : 98% Smile code : O=C1NC2=C(C=CC=C2)C1=O MDL Number : MFCD00005718 MolFormula : C8H5NO2 MolWeight : 147.1308 Available in stock : 134475.895
More details are to be found here
Purity : 98% Smile code : O=C1NC2=C(C=CC=C2)C1=O MDL Number : MFCD00005718 MolFormula : C8H5NO2 MolWeight : 147.1308 Available in stock : 134475.895
More details are to be found here
Purity : 98% Smile code : O=C1NC2=C(C=CC=C2)C1=O MDL Number : MFCD00005718 MolFormula : C8H5NO2 MolWeight : 147.1308 Available in stock : 134475.895
More details are to be found here
Purity : 98% Smile code : O=C1NC2=C(C=CC=C2)C1=O MDL Number : MFCD00005718 MolFormula : C8H5NO2 MolWeight : 147.1308 Available in stock : 134475.895
More details are to be found here
More details are to be found here
Product Name: Isatin
Synonyms: TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
CAS: 91-56-5
MF: C8H5NO2
MW: 147.13
EINECS: 202-077-8
mp 193-195 °C (dec.)(lit.)
Fp 220 °C
storage temp. Store at RT.
Decomposition 194 ºC
Merck 14,5104
BRN 383659
Stability: Stable. Incompatible with strong acids.
chemical property: orange-red powder or crystals
Uses
Indigo and its derivatives as indigo and related dyes and some of the raw materials for the preparation of drugs. Because of its hepatotoxicity, indigo derivatives are no longer used in the pharmaceutical industry.
A precipitant for the detection of ketones and silver. Spectrophotometric Determination of Thiophene, Thiol, Thiophene, Urine Blue and Proline.
Preparation Source
By indophenol or indigo by oxidation and prepared. Indigo was first isolated by the French chemist Auguste Laurent. In 1878, Adolf von Bayer completed the full synthesis of isatin. In 1880, Bayer developed a method of synthesizing isatin from o-nitrocinnamic acid. In 1883, from the o-nitrobenzaldehyde synthesis of isatin method by Bayer patent. Since then, indigo as the raw material for the synthesis of indigo gradually replace the method of extraction from plants, as the main source of indigo.
Product Name: Isatin
Synonyms: TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
CAS: 91-56-5
MF: C8H5NO2
MW: 147.13
EINECS: 202-077-8
mp 193-195 °C (dec.)(lit.)
Fp 220 °C
storage temp. Store at RT.
Decomposition 194 ºC
Merck 14,5104
BRN 383659
Stability: Stable. Incompatible with strong acids.
chemical property: orange-red powder or crystals
Uses
Indigo and its derivatives as indigo and related dyes and some of the raw materials for the preparation of drugs. Because of its hepatotoxicity, indigo derivatives are no longer used in the pharmaceutical industry.
A precipitant for the detection of ketones and silver. Spectrophotometric Determination of Thiophene, Thiol, Thiophene, Urine Blue and Proline.
Preparation Source
By indophenol or indigo by oxidation and prepared. Indigo was first isolated by the French chemist Auguste Laurent. In 1878, Adolf von Bayer completed the full synthesis of isatin. In 1880, Bayer developed a method of synthesizing isatin from o-nitrocinnamic acid. In 1883, from the o-nitrobenzaldehyde synthesis of isatin method by Bayer patent. Since then, indigo as the raw material for the synthesis of indigo gradually replace the method of extraction from plants, as the main source of indigo.
Product Name: Isatin
Synonyms: TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
CAS: 91-56-5
MF: C8H5NO2
MW: 147.13
EINECS: 202-077-8
mp 193-195 °C (dec.)(lit.)
Fp 220 °C
storage temp. Store at RT.
Decomposition 194 ºC
Merck 14,5104
BRN 383659
Stability: Stable. Incompatible with strong acids.
chemical property: orange-red powder or crystals
Uses
Indigo and its derivatives as indigo and related dyes and some of the raw materials for the preparation of drugs. Because of its hepatotoxicity, indigo derivatives are no longer used in the pharmaceutical industry.
A precipitant for the detection of ketones and silver. Spectrophotometric Determination of Thiophene, Thiol, Thiophene, Urine Blue and Proline.
Preparation Source
By indophenol or indigo by oxidation and prepared. Indigo was first isolated by the French chemist Auguste Laurent. In 1878, Adolf von Bayer completed the full synthesis of isatin. In 1880, Bayer developed a method of synthesizing isatin from o-nitrocinnamic acid. In 1883, from the o-nitrobenzaldehyde synthesis of isatin method by Bayer patent. Since then, indigo as the raw material for the synthesis of indigo gradually replace the method of extraction from plants, as the main source of indigo.
Product Name: Isatin
Synonyms: TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
CAS: 91-56-5
MF: C8H5NO2
MW: 147.13
EINECS: 202-077-8
mp 193-195 °C (dec.)(lit.)
Fp 220 °C
storage temp. Store at RT.
Decomposition 194 ºC
Merck 14,5104
BRN 383659
Stability: Stable. Incompatible with strong acids.
chemical property: orange-red powder or crystals
Uses
Indigo and its derivatives as indigo and related dyes and some of the raw materials for the preparation of drugs. Because of its hepatotoxicity, indigo derivatives are no longer used in the pharmaceutical industry.
A precipitant for the detection of ketones and silver. Spectrophotometric Determination of Thiophene, Thiol, Thiophene, Urine Blue and Proline.
Preparation Source
By indophenol or indigo by oxidation and prepared. Indigo was first isolated by the French chemist Auguste Laurent. In 1878, Adolf von Bayer completed the full synthesis of isatin. In 1880, Bayer developed a method of synthesizing isatin from o-nitrocinnamic acid. In 1883, from the o-nitrobenzaldehyde synthesis of isatin method by Bayer patent. Since then, indigo as the raw material for the synthesis of indigo gradually replace the method of extraction from plants, as the main source of indigo.
Product Name: Isatin
Synonyms: TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
CAS: 91-56-5
MF: C8H5NO2
MW: 147.13
EINECS: 202-077-8
mp 193-195 °C (dec.)(lit.)
Fp 220 °C
storage temp. Store at RT.
Decomposition 194 ºC
Merck 14,5104
BRN 383659
Stability: Stable. Incompatible with strong acids.
chemical property: orange-red powder or crystals
Uses
Indigo and its derivatives as indigo and related dyes and some of the raw materials for the preparation of drugs. Because of its hepatotoxicity, indigo derivatives are no longer used in the pharmaceutical industry.
A precipitant for the detection of ketones and silver. Spectrophotometric Determination of Thiophene, Thiol, Thiophene, Urine Blue and Proline.
Preparation Source
By indophenol or indigo by oxidation and prepared. Indigo was first isolated by the French chemist Auguste Laurent. In 1878, Adolf von Bayer completed the full synthesis of isatin. In 1880, Bayer developed a method of synthesizing isatin from o-nitrocinnamic acid. In 1883, from the o-nitrobenzaldehyde synthesis of isatin method by Bayer patent. Since then, indigo as the raw material for the synthesis of indigo gradually replace the method of extraction from plants, as the main source of indigo.
Detailed information on the suppliers of
Isatin |
Properties:
... more properties and specification on Isatin
|
|
Privileged suppliers
Last update 2024-06-12
|


 details
details details
details details
details







